Alzheimer’s Disease and Dementia Research Highlights From 2018
The world of Alzheimer’s disease and dementia research is one of constant evolution. Scores of professionals are perpetually at work looking for new ways to prevent, diagnose, and treat these diseases. This tireless ingenuity is both a testament to the dedication and innovation that supports the field, as well as an obvious necessity, given the difficulty that has been experienced in finding effective preventions and treatments for these conditions throughout history.
Thankfully, due to a large amount of ongoing research, there is always hope that researchers will soon make a breakthrough. Each year brings significant advancements in the fight against Alzheimer’s disease and other dementias, and 2018 was no exception.
Highlights from 2018’s Alzheimer’s Disease and Dementia Research Findings
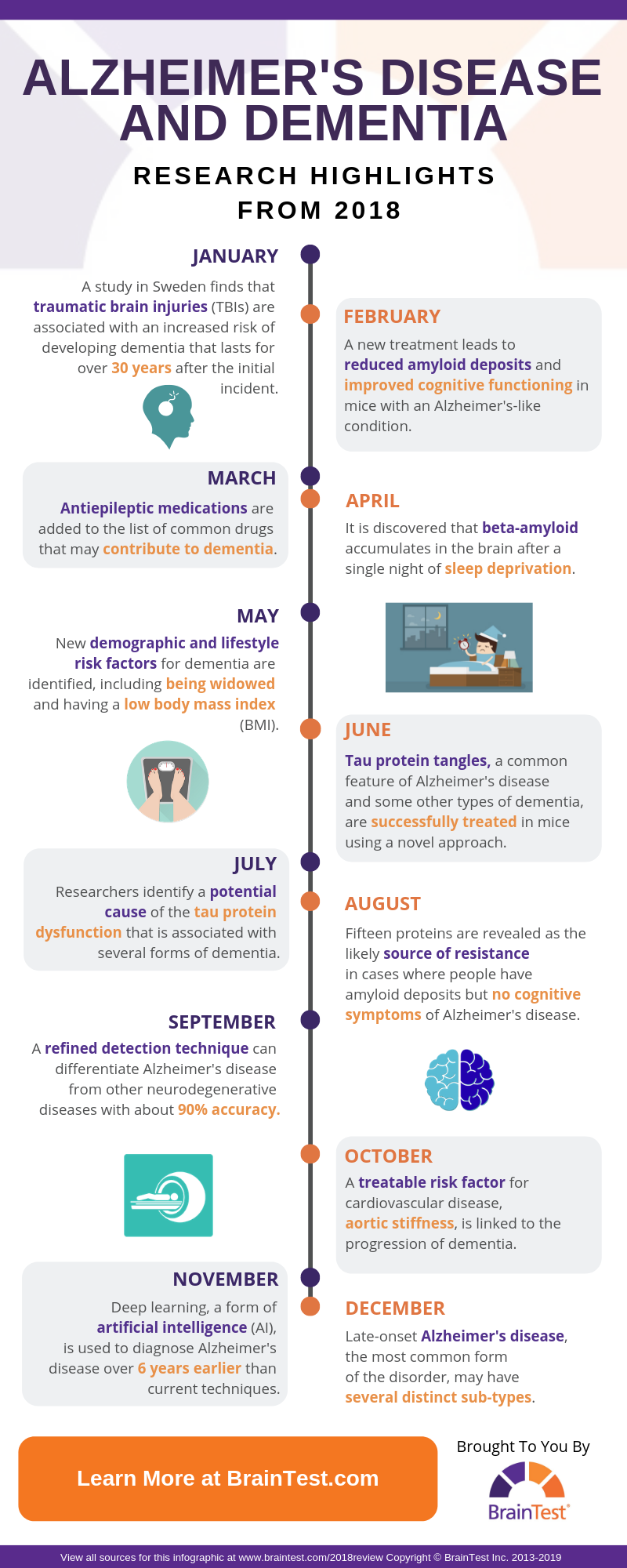
January
It has long been recognized that traumatic brain injury (TBI) is associated with an increased risk of getting dementia, but details about the relationship are still being uncovered. A study published in the journal PLOS Medicine startlingly suggests that this susceptibility can last for more than 30 years after the injury takes place.
The researchers examined data from more than 3.3 million Swedish individuals that were over the age of 50. Groups of people that had a TBI were compared to those who had not. Their analysis revealed a significant association between having a TBI and dementia. The risk did decrease over time but was still observable over 30 years later.
February
Beta-amyloid is a protein that is highly associated with Alzheimer’s disease. It forms clumps in the brains of people with the disease. These deposits are called plaques and are a significant source of the damage that is caused by the condition. It is still unclear if the abnormal accumulation of beta-amyloid is a cause of Alzheimer’s disease or if it is a symptom.
There has historically been little success in developing treatments that directly attack the beta-amyloid deposits, but this streak may soon come to an end. Researchers working with mice reported success in significantly reducing beta-amyloid deposits using a new treatment method. Instead of attacking beta amyloid directly, they targeted a specific enzyme that is required for constructing the protein. Most importantly, the treatment also resulted in better cognitive functioning.
March
One of the most complex aspects of treating dementia is finding a combination of medications that effectively reduce the many symptoms without also causing harm. Dementia patients may be candidates for a wide variety of medications due to the disease itself or co-existing conditions. Unfortunately, many potentially helpful medications (like those that combat anxiety and psychosis) are also known to exacerbate cognitive decline in dementia patients. It now appears that anti-epileptic drugs (for seizures) can be added to this list.
Researchers analyzed data from the Finnish health register and German health insurance by identifying people with dementia (over 90,000 in total) and comparing them with a matched group who did not. They found that the use of anti-epileptic medications was linked with a significantly higher risk of developing dementia.
Due to the design of the study, it cannot be said that these drugs are a cause of dementia (the results could be due to an uncontrolled influence since the data was not obtained with this analysis in mind), but it does set the stage for future researchers to clarify the relationship between anti-epileptic drugs and dementia.
April
Although it is most commonly associated with Alzheimer’s disease, the protein beta-amyloid is actually not rare to find in a normal human brain. More specifically, it tends to be located in the interstitial fluid (ISF), which fills the spaces between brain cells.
The function of beta-amyloid is not clear if it even has one at all (some researchers theorize that the protein is simply a waste product of metabolism). Beta-amyloid is regularly removed from the brain via the immune system, but new research shows that the process may be impaired by a single night of sleep deprivation.
Alzheimer’s disease is accompanied by abnormal deposits of beta-amyloid, but it is not known how the clumps are formed. The discovery that sleep deprivation inhibits beta-amyloid removal from the brain may provide a new pathway to understanding the development of amyloid plaques. It also indicates that sleep loss may be a considerable risk factor for developing Alzheimer’s disease, though more research will be needed to determine if excess beta-amyloid due to sleep deprivation is directly linked to the condition.
May
The identification of risk factors for dementia is a key aspect of prevention research. It serves two main purposes:
- People can be advised to manage any risk factors that they can control.
- Studying risk factors may lead to the ultimate discovery of a cause for Alzheimer’s disease and other forms of dementia.
A large-scale study completed in Massachusetts has succeeded in detecting multiple potential risk factors for dementia that were not previously recognized. Being a widow, having a low body mass index (BMI), and getting less sleep during midlife were all associated with a higher risk of developing dementia. While there is little a person can do to avoid being widowed, BMI and sleep habits can usually be modified with personal training and/or therapeutic methods.

Image via Pixabay
June
Beta-amyloid is not the only protein that is linked to dementia. Tau is an important bodily protein that helps to support the structure of cells. In Alzheimer’s disease and some other forms of dementia, abnormal tau separates from cells and creates damaging tangles between brain cells. Like beta-amyloid, tau has long been a focus of dementia research and has remained resistant to treatment attempts.
In 2018, researchers reported their apparent success in reducing the amount of tau in the brains of mice that had a condition similar to Alzheimer’s disease (also known as an “animal model”). The effectiveness of the treatment is attributed to the targeting of an enzyme that helps create tau, which is a different approach than the traditional method of targeting the tau itself. Future trials are planned to evaluate its impact on humans.
July
A multi-institute research effort appears to have made significant strides in determining the cause of tau protein abnormalities that are found in Alzheimer’s disease and other types of dementia. The science behind the finding is quite complicated and relates to the way in which tau protein is created in the body.
In essence, some tau abnormalities seem to be the product of a faulty “template” for the protein’s production. If researchers can find a way to prevent these templates from being corrupted, then they may also be able to inhibit the formation of tau tangles and similar obstructions.
August
One of the most mystifying discoveries in Alzheimer’s research is the fact that some people have the abnormal protein formations that signify the condition but lack any cognitive symptoms. This apparent resistance is of great interest to professionals who are diligently working toward finding effective treatments for the disease. In 2018, researchers may have made a critical discovery in determining the source of their resistance.
Previous to this new finding, it was known that at least part of the reason for this symptom resistance was that the protein plaques and tangles, though present, did not accumulate in the synapses of neurons as they do in typical Alzheimer’s cases. Synapses are the spaces between neurons where communication occurs, usually via chemical transmission.
In this study, researchers identified 15 specific proteins at the synapses of patients who have plaques and tangles without cognitive symptoms. These proteins are not present in people who have the typical cognitive symptoms of Alzheimer’s disease, and thus may represent a key defensive mechanism.
September
Differential diagnosis is the process that professionals use to narrow down the potential causes of symptoms in an individual, and it is an important part of identifying neurodegenerative diseases like the multiple types of dementia. It can be a challenge to correctly diagnose a neurodegenerative illness, especially when multiple options feature similar symptoms (such as the tau tangles found in Alzheimer’s disease and some other forms of dementia).
This past September, research published in the academic journal JAMA details a new advancement using positron emission tomography (PET) that may vastly improve the ability to differentially diagnose Alzheimer’s disease from other neurodegenerative conditions. The study included over 700 participants with varying forms of neurodegeneration and reported a success rate near 90% in correctly identifying Alzheimer’s disease.
October
Cardiovascular problems are historically linked with the development of dementia, but researchers are still trying to better understand the connection. It is extremely desirable to gain insight into how cardiovascular issues related to dementia, as they can often be treated with medication and lifestyle changes.
Finding a direct connection between dementia and cardiovascular conditions may allow for treatment, or even prevention, of Alzheimer’s disease and other forms of dementia by focusing on alleviating cardiovascular problems. In this study, researchers determined that an overly thick aorta (the largest artery in the body) is linked with an increased risk of developing dementia, and aortic thickness is treatable with medication.
November
Artificial intelligence (AI) has opened up a new world of possibilities for researchers in just about every field. Deep learning is an advanced form of AI that can find patterns in data that even the most highly trained professionals struggle to comprehend. These findings can then be used to make improvements to technology, treatment methods, and more.
This past year, researchers used deep learning to significantly improve the ability of PET scans to detect Alzheimer’s disease at an early stage in its development. Early diagnosis is critical to ensure the best possible outcomes for Alzheimer’s patients, and this advancement may make it possible to detect the formation of the disease more than six years before a final diagnosis could be made by current techniques. As it stands, the best way for a person to receive an early diagnosis is through professional cognitive examinations, such as the SAGE test on which the BrainTest® app is based.
December
Alzheimer’s disease is generally recognized as having two distinct types: early-onset and late-onset. Early onset Alzheimer’s disease occurs at a younger age (prior to 65 years, often in the 40s or 50s) and is by far the most uncommon form of the disease, making up only about 5% of all cases. Late-onset Alzheimer’s is usually referred to simply as Alzheimer’s disease and is considered to be a single condition, even though it can vary wildly in progression and symptoms on a case-to-case basis.
A new study may greatly expand the current classification system for late-onset Alzheimer’s disease. Researchers examined genetic and cognitive data from five previous studies with a total of over 4,000 participants who had late-onset Alzheimer’s. Their findings suggest that there are in the range of 30 distinct sub-types of the disease, raising the possibility of designing targeted diagnosis, prevention, and treatment methods for each group.
On to 2019

Image via Pixabay
As we can see from this small sample of highlights, 2018 brought about many potentially important research findings in the battle against Alzheimer’s disease and other types of dementia. Given that there are dozens of intense investigations being conducted at any given moment, we can expect that these advancements will continue into the new year. With these efforts, and perhaps a little luck, one of these years will play host to a major breakthrough that brings immediate benefits to the population. Could that year be 2019? Stay tuned.
Click here to view all sources for this review.